In the field of metallurgy, extracting metals from their ores involves several important processes. One such key process is calcination, which plays a vital role in preparing ores for further extraction. Calcination is defined as the process of heating an ore in the absence or limited supply of air. This process helps remove unwanted substances and convert the ore into a more suitable form for metal extraction. Understanding calcination is essential for students studying chemistry, especially in topics related to metallurgy and industrial processes.
What is Calcination?
Calcination is a thermal process in which an ore is heated strongly below its melting point without allowing excess air. During this process, volatile impurities such as moisture, carbon dioxide, and organic materials are removed. It is mainly used for ores that are carbonates or hydrated in nature.
For example, when calcium carbonate (limestone) is heated, it decomposes into calcium oxide (quicklime) and carbon dioxide gas. This reaction is a classic example of calcination.
Chemical Reactions in Calcination
Calcination involves decomposition reactions. A common example is:
Calcium Carbonate → Calcium Oxide + Carbon Dioxide
This reaction shows how heating in the absence of air leads to the breakdown of a compound into simpler substances. The removal of gases like CO₂ makes the ore more porous and easier to process in the next steps.
Why is Calcination Important?
1. Removal of Moisture
Many ores contain water molecules. Calcination removes this moisture, making the ore dry and suitable for further processing.
2. Elimination of Volatile Impurities
Impurities like carbon dioxide and organic matter are driven off during heating, purifying the ore.
3. Conversion into Oxides
Calcination converts carbonate and hydroxide ores into oxides, which are easier to reduce and extract metals from.
4. Improves Efficiency of Extraction
By making the ore porous and free from impurities, calcination increases the efficiency of subsequent metallurgical processes.
Calcination vs Roasting
Calcination is often compared with another process called roasting, but they are different:
- Calcination: Heating in absence or limited air
- Roasting: Heating in the presence of excess air or oxygen
While calcination removes volatile substances, roasting is mainly used to convert sulfide ores into oxides by oxidation.
Types of Ores Treated by Calcination
Calcination is mainly used for:
- Carbonate ores (e.g., calcium carbonate)
- Hydrated ores (e.g., aluminum hydroxide)
These ores decompose easily when heated, making calcination an ideal method.
Industrial Applications
Calcination is widely used in industries such as:
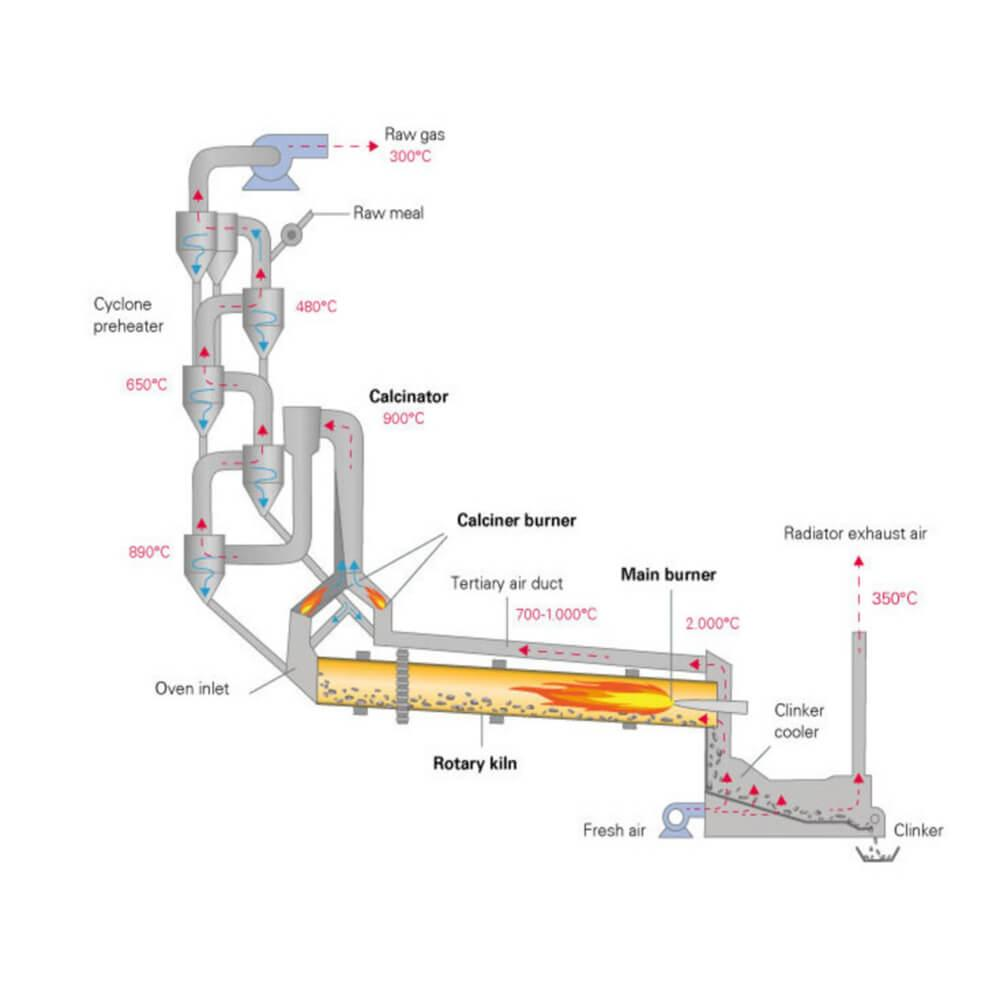
- Cement Industry: Limestone is calcined to produce lime, an important component of cement
- Metallurgy: Used in extraction of metals like zinc and aluminum
- Chemical Industry: Used in production of various oxides and chemicals
The process is usually carried out in furnaces or rotary kilns, where temperature and air supply are carefully controlled.
Environmental Considerations
Although calcination is useful, it releases gases like carbon dioxide, which contribute to environmental pollution. Modern industries are working on technologies to reduce emissions and make the process more eco-friendly.
Advantages of Calcination
- Removes impurities effectively
- Converts ores into useful forms
- Improves metal extraction efficiency
- Simple and widely used process
Limitations of Calcination
- Requires high temperatures
- Releases CO₂, contributing to pollution
- Not suitable for all types of ores
Calcination is an important step in the metallurgical process, involving the heating of ores in the absence or limited supply of air. It helps remove impurities, convert ores into oxides, and prepare them for metal extraction. Despite some environmental challenges, calcination remains a widely used and essential process in industries such as cement production and metallurgy. By understanding calcination, students can gain deeper insight into how raw materials are transformed into useful metals and products that we use in everyday life.
For More Information and Updates, Connect With Us
- Name Survi kumari
- Phone Number: +91-7488713635
- Email ID: survi.eeplclassroom@gmail.com
- Our Platforms:
- Digilearn Cloud
- EEPL Test
- Live Emancipation
- Follow Us on Social Media:
- Instagram – EEPL Classroom
- Facebook – EEPL Classroom
Stay connected and keep learning with EEPL Classroom!

Leave a Reply