Reduction is a fundamental concept in chemistry, especially in the study of chemical reactions. It is commonly defined as the addition of hydrogen, removal of oxygen, or gain of electrons from a substance. Understanding reduction is essential because it is always paired with another process called oxidation. Together, they form what is known as a redox (reduction-oxidation) reaction.
What is Reduction?
Reduction refers to a chemical process in which a substance undergoes a change that leads to a decrease in its oxidation state. In simple terms, reduction can be understood in three main ways:
- Addition of hydrogen to a substance
- Removal of oxygen from a substance
- Gain of electrons
These definitions are interconnected and help explain how substances transform during chemical reactions.
Reduction as Addition of Hydrogen
One of the easiest ways to understand reduction is by looking at the addition of hydrogen. When a substance gains hydrogen atoms, it is said to be reduced.
For example, consider a reaction where hydrogen is added to a compound:
- A substance combines with hydrogen
- Its chemical structure changes
- It becomes more “reduced”
This type of reaction is commonly seen in organic chemistry, especially in processes like hydrogenation.
Reduction as Removal of Oxygen
Another way to define reduction is the removal of oxygen from a substance. If a compound loses oxygen atoms during a reaction, it is undergoing reduction.
A simple example:
- Metal oxide → Metal + Oxygen
Here, the metal oxide loses oxygen and becomes a pure metal. This process is widely used in industries, such as extracting metals from their ores.
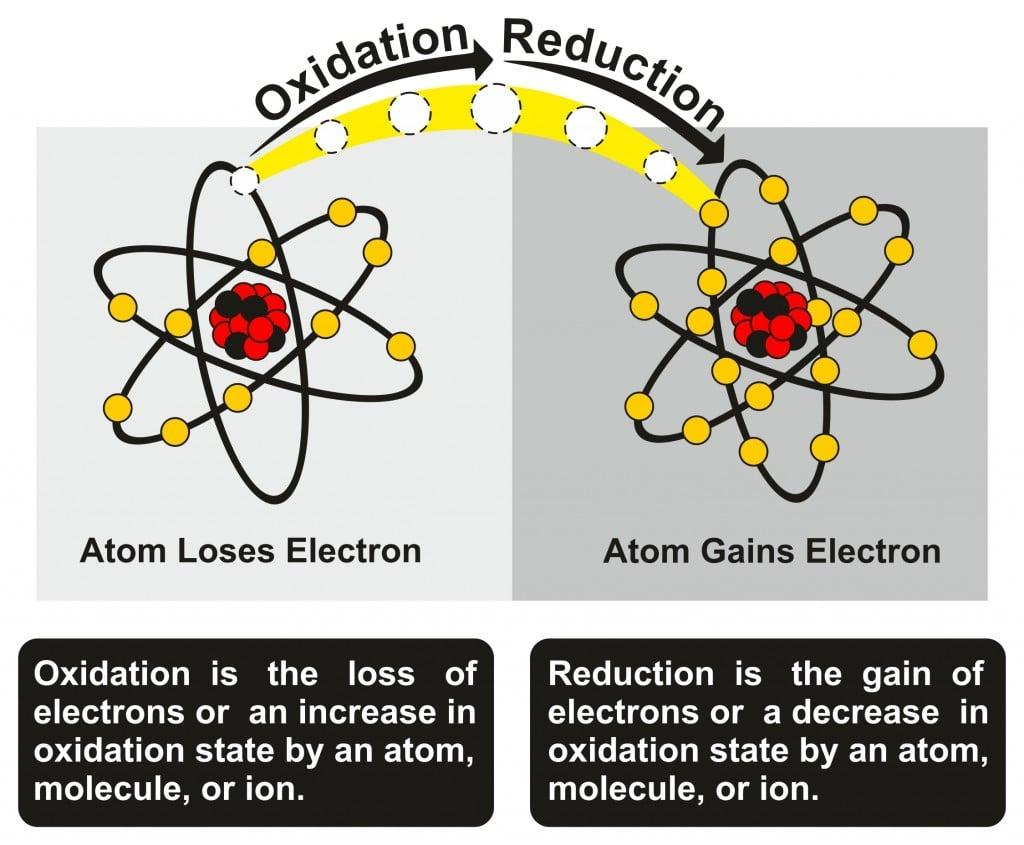
Reduction as Gain of Electrons
In modern chemistry, reduction is most accurately defined as the gain of electrons. When a substance gains electrons, its oxidation state decreases, meaning it is reduced.
For example:
- Cu²⁺ + 2e⁻ → Cu
In this reaction, copper ions gain electrons and are reduced to metallic copper.
Relationship Between Oxidation and Reduction
Reduction cannot occur alone. It always happens alongside oxidation. While one substance is reduced, another is oxidized.
- Oxidation: Loss of electrons, addition of oxygen, or removal of hydrogen
- Reduction: Gain of electrons, addition of hydrogen, or removal of oxygen
This simultaneous process is called a redox reaction.
Everyday Examples of Reduction
Reduction is not just a theoretical concept; it occurs in everyday life and industrial processes:
1. Rust Removal
When rust (iron oxide) is converted back into iron, oxygen is removed. This is a reduction process.
2. Photosynthesis
Plants reduce carbon dioxide into glucose by adding hydrogen during the process.
3. Metal Extraction
In metallurgy, metals like iron are extracted from their ores by removing oxygen.
4. Fuel Combustion
In some stages of fuel reactions, reduction processes help in energy production.
Importance of Reduction
Reduction plays a vital role in both natural and industrial processes:
- It helps in the production of metals
- It is essential for energy transfer in cells
- It is involved in biochemical reactions like respiration and photosynthesis
- It supports many industrial chemical processes
Without reduction, many essential reactions in nature and technology would not occur.
Easy Way to Remember Reduction
A popular way to remember reduction is:
“OIL RIG”
- Oxidation Is Loss (of electrons)
- Reduction Is Gain (of electrons)
You can also remember:
- Reduction = Gain of electrons
- Reduction = Addition of hydrogen
- Reduction = Removal of oxygen
Reduction is a key concept in chemistry that describes how substances gain electrons, add hydrogen, or lose oxygen during reactions. It is always linked with oxidation, forming redox reactions that drive countless natural and industrial processes.
By understanding reduction, students can better grasp how chemical reactions work and how they impact the world around us. Whether it’s extracting metals, producing energy, or supporting life processes, reduction plays a crucial role in maintaining the balance of chemical systems.
For More Information and Updates, Connect With Us
- Name Survi kumari
- Phone Number: +91-7488713635
- Email ID: survi.eeplclassroom@gmail.com
- Our Platforms:
- Digilearn Cloud
- EEPL Test
- Live Emancipation
- Follow Us on Social Media:
- Instagram – EEPL Classroom
- Facebook – EEPL Classroom
Stay connected and keep learning with EEPL Classroom!

Leave a Reply